African swine fever
African swine fever (ASF) is a highly contagious viral disease of domestic and wild pigs that may result in high or low case mortality rates, fever, hyperaemia of the skin and a variety of other clinical signs, including incoordination, diarrhoea and pneumonia. Due to its high economic burden and the lack of a commercially available vaccine, it is considered one of the most important diseases of pigs worldwide and is listed as a notifiable disease by the World Organisation for Animal Health (OIE) Terrestrial Animal Health Code.
ASF has never been reported in Australia and an outbreak of ASF would have significant impacts on productivity and international market access. The disease would also likely be difficult and costly to eradicate. The pork industry is an important contributor to Australian livestock production and the national economy, and existing import controls, applied since 2004 in most cases, include measures to manage the risk from ASF in pig meat and porcine products. These are supported by internal controls (such as the national swill feeding ban) over the use of products within Australia that contain imported porcine-derived ingredients. These internal controls manage the risk of susceptible livestock being exposed to exotic livestock diseases.
ASF was limited to sub-Saharan Africa until 1957 when the disease occurred in Portugal. The disease was then reported from the 1960s to 1990s in Belgium, Brazil, Cuba, the Dominican Republic, France, Haiti, Italy, Malta, the Netherlands, Portugal and Spain. The disease has since been eradicated from most of these countries but remains endemic in feral pigs in Sardinia, an island of Italy.
Outbreaks have occurred since June 2007 in many countries—most notably the Ukraine, western Russia, and the EU (member states in central and eastern Europe)—where the disease had not previously been reported. Control efforts in these countries have not to date effectively prevented the spread of this disease. Most recently, Belgium confirmed two cases of ASF in their wild boar population on 13 September 2018, the first known occurrence of the disease in Western Europe since the virus landed in Georgia and Armenia in 2007.
ASF was also reported for the first time in China in August 2018 and, given the experience with ASF in other countries, eradication in China will likely require significant resourcing and control measures. Should the disease fail to be controlled, food security in China may be impacted.
Almost half the world’s pig production occurs in China, and an estimated 500 million pigs are slaughtered per annum. Also, China has a large population of wild boar able to harbour the disease without showing clinical signs of disease. In addition, around 52 percent of Chinese pig herds are raised in backyard farms, which makes disease containment, tracing and diagnosis difficult.
At the time of writing of this bulletin (14 September 2018), ASF in China continues to spread with reported outbreaks in six provinces and more than 40,000 pigs culled. The transport of pigs and related products has been banned in affected and surrounding areas but the origin of the disease and the mechanism of transmission have not yet been identified. The rapid spread and widespread nature of the reported cases has led to the Food and Agriculture Organization of the United Nations expressing concerns about the risk of spread to other parts of Asia.
Aetiology
The causative agent of ASF is African swine fever virus (ASFV), an enveloped DNA virus of the genus Asfivirus, family Asfarviridae. The virus is very resistant to inactivation in the environment and virus isolates vary greatly in their virulence. In Australia, both domestic and feral pigs (Sus scrofa) are susceptible hosts for ASF. Wild pigs have been recognised as sources of infection in a number of outbreaks in endemic countries and, should ASF enter Australia, the feral pig population may act as a reservoir of the virus.
Epidemiology
ASFV spreads rapidly via both direct and indirect pathways and the incubation period is usually between 4 and 19 days.
Pigs with acute disease shed virus in high concentrations in all secretions and excretions, particularly those that contain blood. Infection by the oral and respiratory routes can also occur between pigs in close contact. Pigs that survive natural infection may develop antibodies against ASFV from 7 to 10 days post-infection and these antibodies can persist for long periods of time. Movement of infected pigs is the most important method of spread between piggeries.
Virus is transmitted principally by direct contact with infected pigs, while indirect transmission occurs via contact with fomites, such as equipment and personnel, or following ingestion of infected pig meat or products. The feeding of food scraps or food waste that contains or has come into contact with meat or meat products (swill feeding) is illegal in Australia, to prevent a range of diseases. The carcasses of infected pigs have also been linked to the spread of ASF. This may be an important pathway for spread of ASF between feral pig populations.
ASFV replicates in Ornithodoros ticks and is transmitted to swine through the bite of the tick, which plays an important role in transmission in Africa. The potential role of Ornithodoros and other ticks in Australia is not known.
Clinical disease
Disease severity varies greatly and is influenced by host age, herd immunity and the strain and virulence of ASFV encountered. Pigs infected with ASFV may develop acute, chronic, or subclinical disease, and the clinical signs and case mortality rates are extremely variable. In general, young naive animals are more severely affected, but the emergence of less virulent ASFV strains and milder clinical disease reinforces the need for vigilant testing and reporting of all suspect cases.
Clinical signs of acute ASF may include, but are not limited to: pyrexia (39.5 – 42˚C), inappetance, gastrointestinal and neurological abnormalities, abortion or foetal deformities, conjunctivitis, nasal discharge, and skin haemorrhages and cyanosis (particularly on extremities such as the ears and snout). Case fatality rates can reach 100% following acute highly virulent disease. However, animals with chronic ASF may present with milder clinical signs, including: general ill-thrift, fluctuating pyrexia, pneumonia, alopecia and dermatitis. In the subclinical form of the disease, animals may become chronic carriers without overt clinical signs.
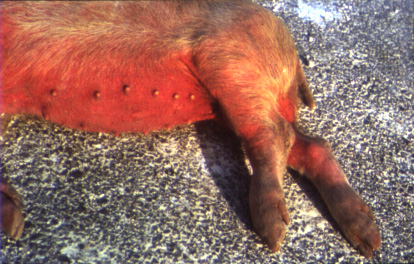
(Source: P.J. Wilkinson.)
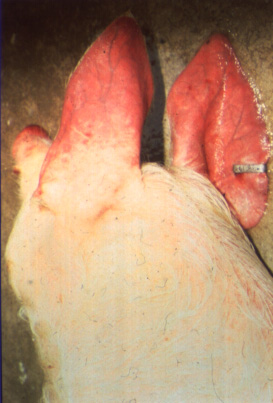
(Source: P.J. Wilkinson.)
Pathology
The most common post-mortem findings in ASF-positive pigs are signs of haemorrhage, such as: petechiae on the kidneys, bladder and lymph nodes; splenic enlargement; gastric ulceration; and pneumonia, pleuritis or bronchitis. The acute form may present with more profound haemorrhaging across all organs, fluid in the body cavities and lungs and pyramidal infarcts along the margin of the spleen. Gross pathology of chronic ASF varies, and is generally indicative of a more prolonged illness. Lesions may include fibrinous pericarditis, pleurisy, button ulcers of the large intestine at the ileocaecal valve, lobular lung consolidation, and thymic atrophy.
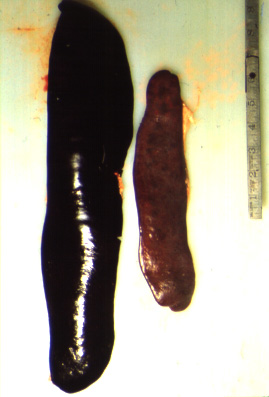
(Source: C.A. Mebus.)
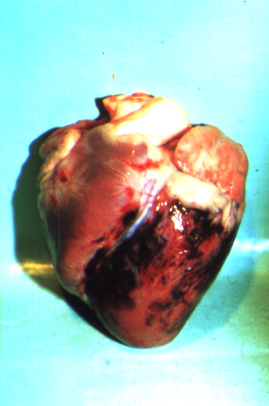
(Source: P.J. Wikinson.)
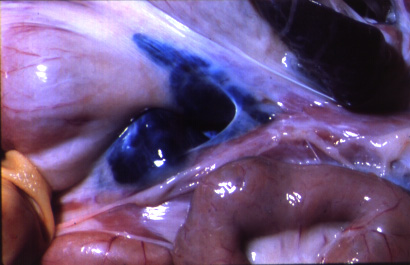
(Source: C.A. Mebus.)
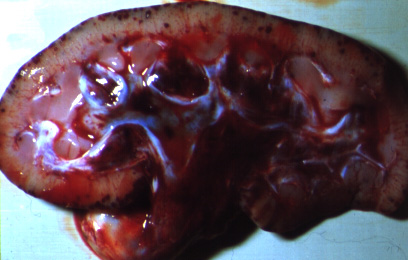
(Source: C.A Mebus.)
Diagnosis
As clinical signs and post-mortem lesions are not unique to ASF, suspicion of the disease must be confirmed by laboratory diagnosis. Several pigs (at least five if possible) should be submitted for post-mortem examination, as there may be great variability in lesions presented in individual animals. A composite picture of all lesions seen should be recorded.
Samples should be taken from affected (especially pyrexic) pigs killed immediately before a post-mortem examination, and from pigs that have recently died (including stillborn piglets and aborted foetuses). Collect:
- serum—at least 30 samples from suspected chronically infected animals
- EDTA blood (7-10 ml/animal) from live, clinically affected animals
- fresh tissue from the spleen, lymph nodes (gastro-hepatic, mesenteric and sub-mandibular) tonsils, lung, kidney and ileum (2 g of each tissue); bone marrow may also be useful from dead wild animals that have been dead for some time, as it is likely to be relatively well preserved
- fixed tissue—a full range of tissues (including the brain) in neutral-buffered formalin.
Chill blood samples and unpreserved tissue samples either at 4°C, or with frozen gel packs. Do not freeze samples at -20°C as it reduces the sensitivity when used for virus isolation and molecular diagnostic tests.
Protecting Australia from ASF
In the event of an ASF outbreak, Australia’s policy is to control and eradicate the disease in the shortest possible time using a combination of strategies, including stamping out and movement controls. The Government and Livestock Industry Cost Sharing Deed in Respect of Emergency Animal Disease Responses (EADRA) outlines the government and industry cost-sharing arrangements to fund such a response. Actions taken by Australia in response to an ASF incursion would vary depending on host and agent factors and the extent, location and stage of the outbreak. The Australian Veterinary Emergency Plan (AUSVETPLAN) Disease Strategy for African Swine Fever provides comprehensive control, eradication and post-outbreak surveillance guidelines.
Keeping Australia ASF free
Australia maintains an ASF-free status and greatly reduces the risk of incursion through the enforcement of strict biosecurity policies. Australia’s focus is on ensuring that the level of risk in products that arrive at its borders are already managed to levels that are acceptable. Stringent, scientifically informed import regulations exist for pork and pork products. In addition, swill feeding is illegal under legislation in Australia. At the federal level, the Department of Agriculture, Water and the Environment applies rigorous inspection protocols at Australia’s national borders, and conducts off-shore disease surveillance and risk mitigation activities in Australia’s close neighbours, including Timor-Leste and Papua New Guinea.
The risk of introduction
Illegal importation of ASFV-infected pig products or genetic material remains the most likely source for entry of ASF into Australia. Although swill feeding is prohibited, unlawful or inadvertent feeding of imported infected products to domestic or feral pigs presents the greatest risk, and is believed to be the cause of the first ASF outbreak outside of Africa. Contaminated products may arrive from endemic countries via commercial aircrafts or ships, the international postal service, or waste from fishing vessels. Provided Australia’s modern intensive piggeries continue to practice a high level of biosecurity, the most likely sites of entry for ASF would be smaller commercial or backyard establishments, or scavenging feral pigs.
How Australian veterinarians can help
- Ensure that you and your pig-owning clients are aware of and comply with swill feeding legislation (visit www.farmbiosecurity.com.au/livestock/pigs/feeding-your-pigs/).
- Know the clinical signs of African swine fever and other emergency animal diseases.
- Remember that early detection and laboratory confirmation is critical for a rapid and effective response.
- Report unusual cases of disease where emergency animal diseases are suspected, to state or territory government veterinarians directly or through the Emergency Animal Disease Watch Hotline (1800 675 888).
If you suspect an exotic disease, please contact the Emergency Animal Disease Watch Hotline on 1800 675 888 for advice and assistance.
